港聞更多
中國新聞更多
焦點娛聞
體育報導
今日財經
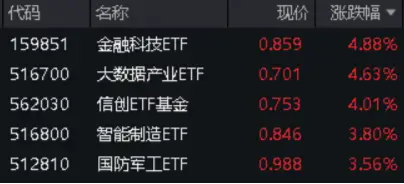
監管發聲,小盤股大反攻!AI概念飆漲,金融科技ETF(159851)反包收漲4.88%!A50ETF華寶(159596)納入兩融標的
今日(4月17日),A股吹響反攻號角,市場一片歡騰雀躍,30個中信一級行業全線飄紅,三大指數集體大漲…
BEAUTY
生活消閒

將軍澳好去處|4大將軍澳拍拖勝地推薦!必去溜冰場、海景Café【同場加映:康城全新人氣樓盤 PARK SEASONS最啱二人世界!】
【將軍澳好去處】想跟另一半享受甜蜜的周末?四大將軍澳拍拖好去處推薦!有溜冰場、海景Cafe、單車館、…











































